| FBA5 | |
| Project ID | 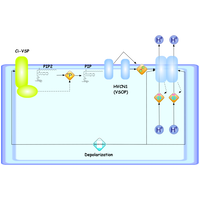 FBA5 [Protein] |
| Project Theme | Structure and function of voltage-sensor domain proteins |
| Project Theme (short) | Voltage-sensor proteins |
| Principal Investigator | Yasushi Okamura |
| Affiliation | Graduate School of Medicine, Osaka University |
| Backgrounds | - We recently discovered the two types of unique voltage-sensing proteins, VSP (voltage sensor-containing phosphatase) and VSOP (voltage sensor only protein) - We will pursue structural and functional elucidation of VSP and VSOP |
| Highlights | - Structures of intracellular enzyme domains of VSP have been determined - Dimeric nature of VSOP channels has been revealed |
| Outline | Ion channels, pore-forming membrane proteins, regulate the flow of ions across the membrane in all cells. They can be classified by gating, i.e. what opens and closes the channels. Voltage-gated ion channels open or close depending on the voltage gradient across the plasma membrane. They are composed of several subunits with a pore domain and a voltage-sensing domain (VSD). VSDs control the gate of the pore. Found along the axon in muscle tissues and at the synapse in neurons, voltage-gated ion channels directionally propagate electrical signals. We recently identified the two types of unique voltage-sensing proteins without the pore domains. VSP (voltage sensor-containing phosphatase) contains enzymatic domain instead of the pore. Its enzymatic activity is regulated by its own voltage-sensor domain. While VSOP (voltage sensor only protein) contain a single VSD but no pore domain, it functions as ion channels. Here, we will pursue structural and functional elucidation of VSP and VSOP. |
| Review | J. Physiol. (Lond.) J. Physiol. (Lond.) Physiology (Bethesda) |
| CSML File | FBA5.csml |